As FDA resumes its routine domestic inspections, facilities are likely to notice a new add-on to the inspection: a Q&A Food Defense “Quick Check.” Are you prepared? In the August FSMA Friday webinar, co-sponsored by SafetyChain, TAG Director of Food Safety, Christopher Snabes presented an overview of the requirements of the FSMA Intentional Adulteration (IA) Rule, new compliance dates, training options … and possible Quick Check questions.
Key takeaways from the webinar include:
- The Rule. Did you know that you are subject to the IA Rule – even if you are exempted from other rules of FSMA?
- Food Defense Training. The rule has a number of training requirements – From Food Defense Awareness, required for all workers, for which a free online course is available. To the much more complex fee-based trainings for qualified individuals, such as Conducting Vulnerability Assessments, which can be taught only by an FSPCA/AFDO-approved Lead Instructor (of which TAG has two), for which participants receive a certificate of training.
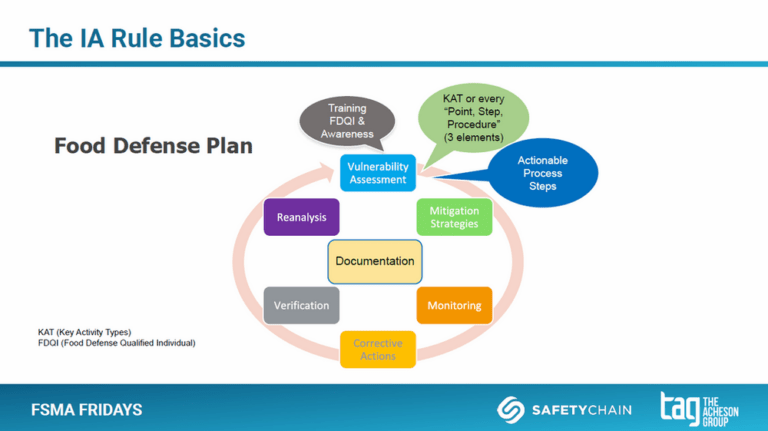
- The Food Defense Plan (FDP). Although FDPs are facility-specific (and required for each facility), there are some very distinct aspects that are required for each FDP. This includes pre-plan prep steps, three vulnerability assessment writing options (based on KATs, 3 Elements, or a hybrid), mitigation strategy development and management, and regular reanalysis.
- The Quick Checks. To “educate while we regulate,” FDA is beginning with Quick Check questions, designed to check the preparedness of facilities. An investigator may ask: Do you have a written FDP? Was it written by an FDQI? Have you identified and KATs? How are you verifying? …
Throughout the presentation Snabes provides expert insight and recommendations on IA Rule compliance, training, writing the FDP, and “passing” a Quick Check.
View the ,webinar replay here, for the full presentation.